Avanos Recalls Catheters after 4 Injuries and 1 Death
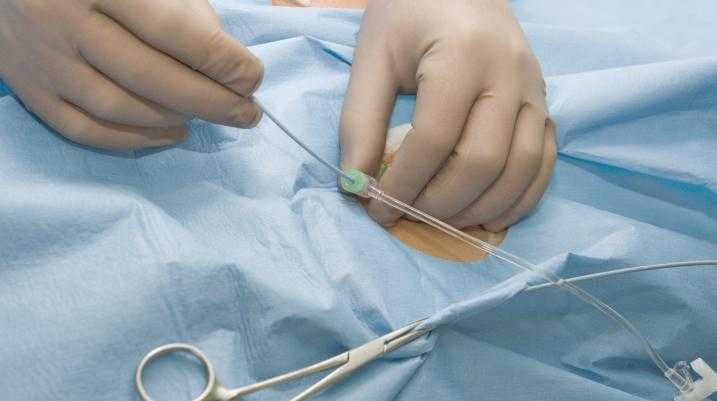
What: The BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Y-Manifold and the BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Elbow Manifold.
Who: Avanos Medical Inc.
Why: The Avanos product has been recalled due to cracked parts that can increase the risk of delayed or prevented ventilation.
How Can I Identify This Recalled Product: Product stock codes for the Y-Manifold can be found here. Product stock codes for the Elbow Manifold can be found here.
What Are My Options: Customers will be contacted by Avanos Customer Service to arrange for the product return, credit, and replacement after the acknowledgment form is submitted.
What Are the BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Y-Manifold or Elbow Manifold?
Distributed from October 20, 2022, to January 23, 2023, BALLARD ACCESS Closed Suction System for Neonates/Pediatrics has two systems that have been recalled. The manifold of the BALLARD ACCESS Closed Suction System is a single-patient-use device that trained medical professionals use to provide access to the artificial airway without breaking the ventilation circuit in children, infants, or neonates. The BALLARD ACCESS Closed Suction manifold is used in conjunction with the BALLARD ACCESS Closed Suction catheter to clear away secretions from the artificial airway of children, infants, and neonates.
What Is the Reason for the Recall?
On February 22, 2023, Avanos Medical Inc. recalled 1,000 of the Avanos BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Y-Manifold or Elbow Manifold. Since the announcement of the recall, FDA has identified this as a Class I recall. A Class I recall is considered the most severe form of recall, and the continued use of the recalled devices can lead to severe injuries or death. The recall happened after Avanos received complaints from its customers over cracked manifolds during use.
Cracked manifolds are dangerous, causing leaks in the respiratory circuit that can lead to the patient receiving inadequate ventilation. If the patient receives inadequate ventilation and oxygenation, it can lead to severe brain damage or even death.
Additional risks from cracked manifolds include introducing a foreign body that could cause the following:
- Infection
- Damage to the airway
- Blocked endotracheal tube
Avanos warns its customers that the continued use of this recalled product could lead to severe injury or death.
Who Is Affected by the Recall?
In the recall, it is noted that those directly affected are children, infants, and neonates who receive care by using a BALLARD ACCESS Closed Suction System. Others affected by the recall include the healthcare personnel providing care for those pediatric or neonatal patients who receive care by using a BALLARD ACCESS Closed Suction System.
Have There Been Any Injuries?
Unfortunately, prior to the recall, there have been four reported injured and one death connected with the BALLARD ACCESS Closed Suction Systems.
What Can Avanos Customers Do?
On February 3, 2023, Avanos Medical Inc. sent their customers an “Urgent Medical Device Recall” letter outlining what they recommended their customers do if in possession of the recalled service devices. In light of the recall, customers are to do the following:
- Check all storage and usage locations for impacted BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Y-Manifold or with Elbow Manifold (labeled “72-hour use”).
- Quarantine all unused impacted products.
For the devices currently in use with patients:
- Check for cracks in the manifold of any product currently in use.
- Replace devices found to have cracked manifolds with a 24-hour use Ballard Closed Suction System for Neonates/Pediatrics.
- Use the 24-hour-use Ballard Closed Suction System for Neonates/Pediatrics for any new placements or scheduled placements.
Customers are expected to complete and return the Acknowledgement Response Form that was included in the letter and send it to FieldActioncare@avanos.com. Emails must include the subject line: FCA-2023-002 Hospital Acknowledgement Response Form.
As previously mentioned, Avanos customers will be contacted by their Customer Service to arrange for the product return, credit, and replacement after the acknowledgment form is submitted.
Should I Contact a Morgan & Morgan Attorney?
Yes. If you or a loved one have experienced an injury or have lost their life after using the BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Y-Manifold or the BALLARD ACCESS Closed Suction System for Neonates/Pediatrics with Elbow Manifold, we may be able to help you.
For more information on how a Morgan & Morgan attorney can help you, please connect with us today by completing our free, no-obligation case evaluation form.
Injured? Getting the compensation you deserve starts here.

Injured?
Not sure what to do next?
We'll guide you through everything you need to know.